Gulab Zode PhD
Lab mission
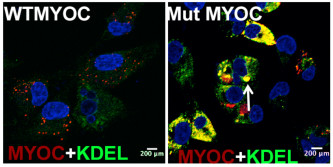
My research focus is to understand how cellular processes including unfolded protein response pathway and autophagy regulate protein misfolding in the glaucomatous trabecular meshwork (TM) and to develop targeted treatments for glaucoma based on the understanding of these pathways. We have developed two novel glaucoma mouse models as well as established various genetic tools to study protein misfolding and biological functions of unfolded protein response pathway and autophagy in glaucoma pathogenesis. We recently discovered that endoplasmic reticulum (ER) stress is associated with trabecular meshwork damage and elevation of intraocular pressure, leading to blindness. We are currently targeting these pathways using genetic and pharmacological manipulations for the treatment of glaucoma.
Projects
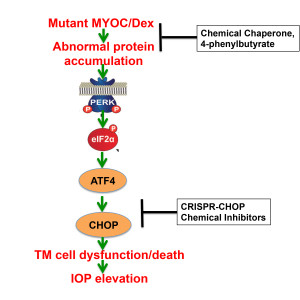
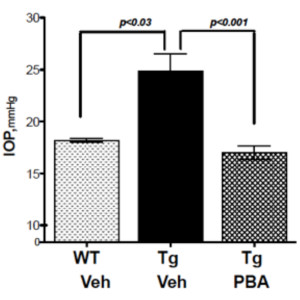
- Small chemical chaperone, sodium 4-phenylbutyrate (PBA) as a treatment for glaucoma: Our previous studies have demonstrated that reducing ER stress via PBA rescues glaucoma in multiple mouse
models of glaucoma. We are currently investigating the molecular mechanisms and therapeutic effectiveness of PBA in reducing elevated IOP using mouse models of glaucoma.
- CRISPR-Cas9 genome editing for treatment of glaucoma. We are currently utilizing CRISPR genome editing technology to knock out various genes involved in the glaucomatous TM damage including Mutant MYOC, ATF4 and CHOP. We also plan to examine whether reduction of these genes via genome editing can rescue mouse models of glaucoma.
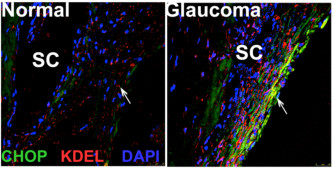
- The role of prodeath ATF4 and CHOP in elevation of IOP: We have recently demonstrated that ER stress-induced prodeath markers ATF-4 and CHOP are increased in the human glaucomatous TM tissues. Our current studies are focused on understanding how induction of ATF4 and CHOP leads to TM dysfunction and IOP elevation.
- To determine how deletion of CHOP rescues glaucoma in mice. Based on our previous studies that demonstrated reduction of CHOP rescues glaucoma in mouse models of glaucoma, we are currently investigating how CHOP rescues glaucoma. We are currently utilizing chemical and genetic approaches to delineate these underlysing molecular mechanisms.
- To determine whether promoting autophagy reduces elevated IOP in mouse models of glaucoma. Based on our preliminary data that suggest comprised autophagy in the glaucomatous TM tissues, we are using various autophagy stimulators to reduce protein misfolding and rescue of glaucoma in mouse models of glaucoma.
Recent Publications
People
- Gulab Zode, PhD (Principle Investigator)
- Ramesh Kasetti, PhD (Post-doctoral fellow)
- Tien Phan (Research Assistant)
- Tzu Chun (Lab Assistant)
Contact
Gulab Zode, PhD
Assistant Professor
North Texas Eye Research Institute
CBH-413
UNT Health Science Center
3500 Camp Bowie Blvd.
Fort Worth, TX 76107
gulab.zode@unthsc.edu
Telephone: 817-735-0360

Social media